§ 799.6756 TSCA partition coefficient (n-octanol/water), generator column method.
(a) Scope—(1) Applicability. This section is intended to meet the testing requirements of the Toxic Substances Control Act (TSCA) (15 U.S.C. 2601).
(2) Source. The source material used in developing this TSCA test guideline is the Office of Pollution Prevention, Pesticides and Toxic Substances (OPPTS) harmonized test guideline 830.7560 (August 1996, final guideline). This source is available at the address in paragraph (e) of this section.
(b)
(1) Purpose. (i) The measurement and estimation of the n-octanol/water partition coefficient (Kow), has become the cornerstone of a myriad of structure-activity relationships (SAR) property. The coefficient has been used extensively for correlating structural changes in drugs with changes observed in biological, biochemical, or toxic effects. These correlations are then used to predict the effect of a new drug for which a Kow could be measured.
(ii) In the study of the environmental fate of organic chemicals, the Kow has become a key parameter. Kow is correlated to water solubility, soil/sediment sorption coefficient, and bioconcentration and is important to SAR.
(iii) Of the three properties that can be estimated from Kow, water solubility is the most important because it affects both the fate and transport of chemicals. For example, highly soluble chemicals become quickly distributed by the hydrologic cycle, have low-sorption coefficients for soils and sediments, and tend to be more easily degraded by microorganisms. In addition, chemical transformation processes such as hydrolysis, direct photolysis, and indirect photolysis (oxidation) tend to occur more readily if a compound is soluble.
(iv) Direct correlations between Kow and both the soil/sediment sorption coefficient and the bioconcentration factor are to be expected. In these cases, compounds that are more soluble in n-octanol (more hydrophobic and lipophilic) would be expected to partition out of the water and into the organic portion of soils/sediments and into lipophilic tissue. The relationship between Kow and the bioconcentration factor, are the principal means of estimating bioconcentration factors. This relationship is discussed in the reference listed in paragraph (e)(14) of this section. These factors are then used to predict the potential for a chemical to accumulate in living tissue.
(v) This section describes a method for determining the Kow based on the dynamic coupled column liquid chromatographic (DCCLC) technique, a technique commonly referred to as the generator column method. The method described herein can be used in place of the standard shake-flask method specified in § 799.6755 for compounds with a log10Kow greater than 1.0.
(2) Definitions. The following definitions apply to this section.
Extractor column is used to extract the solute from the aqueous solution produced by the generator column. After extraction onto a bonded chromatographic support, the solute is eluted with a solvent/water mixture and subsequently analyzed by high-performance liquid chromatography (HPLC), gas chromatography (GC), or any other analytical procedure. A detailed description of the preparation of the extractor column is given in paragraph (c)(1)(i) of this section.
Generator column is used to partition the test substance between the n-octanol and water phases. The column in figure 1 in paragraph (c)(1)(i)(A)(2) of this section is packed with a solid support and is coated with the test substance at a fixed concentration in n-octanol. The test substance is eluted from the column with water and the aqueous solution leaving the column represents the equilibrium concentration of the test substance that has partitioned from the n-octanol phase into the water phase. Preparation of the generator column is described in paragraph (c)(1)(i) of this section.
n-Octanol/water partition coefficient (Kow) is defined as the ratio of the molar concentrations of a chemical in n-octanol and water, in dilute solution. The coefficient Kow is a constant for a given chemical at a given temperature. Since Kow is the ratio of two molar concentrations, it is a dimensionless quantity. Sometimes Kow is reported as the decadic logarithm (log10Kow). In this equation, Coctanol and Cwater are the molar concentration of the solute in n-octanol and water, respectively, at a given temperature. This test procedure determines Kow at 25 ±0.05 °C. The mathematical statement of Kow is:
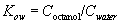
Response factor (RF) is the solute concentration required to give a one unit area chromatographic peak or one unit output from the HPLC recording integrator at a particular recorder and detector attenuation. The factor is required to convert from units of area to units of concentration. The determination of the RF is given in paragraph (c)(3)(iii)(C)(2) of this section.
Sample loop is a 1⁄16 inch (in) outside diameter (O.D.) (1.6 millimeter (mm)) stainless steel tube with an internal volume between 20 and 50 µL. The loop is attached to the sample injection valve of the HPLC and is used to inject standard solutions into the mobile phase of the HPLC when determining the RF for the recording integrator. The exact volume of the loop must be determined as described in paragraph (c)(3)(iii)(C)(1) of this section when the HPLC method is used.
(3) Principle of the test method. (i) This test method is based on the DCCLC technique for determining the aqueous solubility of organic compounds. The development of this test method is described in the references listed in paragraphs (e)(6), (e)(12), and (e)(19) of this section. The DCCLC technique utilizes a generator column, extractor column, and HPLC coupled or interconnected to provide a continuous closed-flow system. Aqueous solutions of the test compound are produced by pumping water through the generator column that is packed with a solid support coated with an approximately 1.0% weight/weight (w/w) solution of the compound in n-octanol. The aqueous solution leaving the column represents the equilibrium concentration of the test chemical which has partitioned from the n-octanol phase into the water phase. The compound is extracted from the aqueous solution onto an extractor column, then eluted from the extractor column with a solvent/water mixture and subsequently analyzed by HPLC using a variable wavelength ultraviolet (UV) absorption detector operating at a suitable wavelength. Chromatogram peaks are recorded and integrated using a recording integrator. The concentration of the compound in the effluent from the generator column is determined from the mass of the compound (solute) extracted from a measured volume of water (solvent). The Kow is calculated from the ratio of the molar concentration of the solute in the 1.0% (w/w) n-octanol and molar concentration of the solute in water as determined using the generator column technique.
(ii) Since the HPLC method is only applicable to compounds that absorb in the UV, an alternate GC method, or any other reliable quantitative procedure must be used for those compounds that do not absorb in the UV. In the GC method the saturated solutions produced in the generator column are extracted using an appropriate organic solvent that is subsequently injected into the GC, or any other suitable analytical device, for analysis of the test compound.
(4) Reference chemicals. (i) Columns 2, 3, 4, and 5 of table 1 in paragraph (b)(4)(ii) of this section list the experimental values of the decadic logarithm of the n-octanol/water partition coefficient (log10Kow) at 25 °C for a number of organic chemicals as obtained from the scientific literature. These values were obtained by any one of the following experimental methods: Shake-flask; generator column; reverse-phase HPLC; or reverse-phase thin-layer chromatography, as indicated in the footnotes following each literature citation. The estimation method of Hawker and Connell as described in paragraph (e)(8) of this section, correlates log10Kow with the total surface area of the molecule and was used to estimate log10Kow for biphenyl and the chlorinated biphenyls. These estimated values are listed in column 7 of table 1 in paragraph (b)(4)(ii) of this section. Recommended values of log10Kow were obtained by critically analyzing the available experimental and estimated values and averaging the best data. These recommended values are listed in column 8 of table 1 in paragraph (b)(4)(ii) of this section.